“Jumping genes” aren't the latest trendy dance move, but they could be the key to better understanding aging. A team of researchers explored how these DNA segments, known for their ability to "jump" from one part of the genome to another, influence longevity. And the results? Surprising.
The research behind hopping genes
- transposable elements (TE), also known as transposons or “jumping genes,” are sequences of DNA that move, or jump, from one location in the genome to another. These movements can sometimes create mutations in the new location, causing a kind of genomic instability that produces aging. Can they be blocked?
The researchers of the Eötvös Loránd University (ELTE) in Hungary they had already published studies in 2015 and 2017, theorizing on how a specific process, called Piwi-piRNA, contributed to aging by helping to control transposable elements. Now, with their latest study, they provide experimental proof of how this process works.
Piwi-piRNA, for friends “P element-induced wimpy testis in Drosophila-Piwi-interacting RNA pathway”, is a specific RNA silencing mechanism that protects genomes from the adverse mutagenic activity of TEs. And it's proving really promising.
The experiment and its discoveries

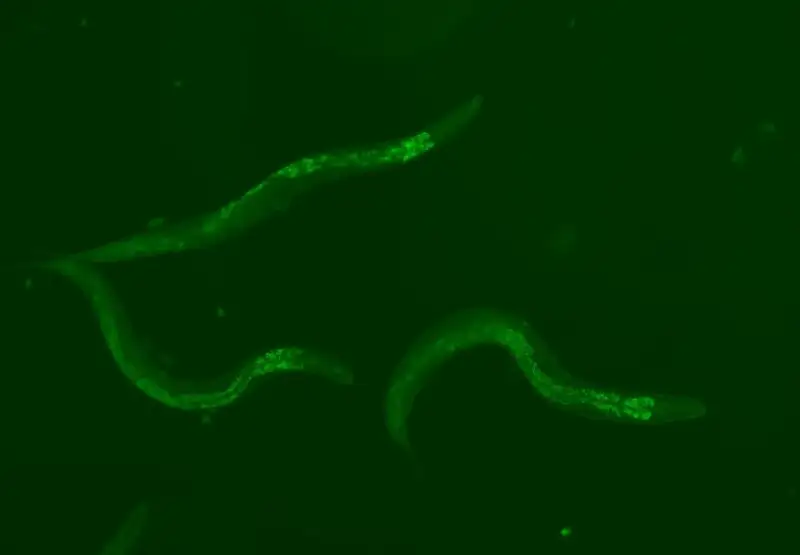
In the latest study, published in the journal Nature Communications. (I link it here) Hungarian researchers conducted experiments on Caenorhabditis elegans. It is a small worm often used as a model for studies on aging, age-related diseases and longevity mechanisms: this is because it has homologs (genes similar in sequence) of about two-thirds of all human disease genes.
By reducing the activity of “jumping gene” families, the researchers found that reducing two specific families, Tc1 and Tc3, the most mobile TEs in the worm, slowed the aging process at different temperatures. At an average of 20°C, lifespan was extended by approximately 10%. By simultaneously reducing both gene families, the life-extending effects are nearly doubled. Reduction of other TE families – Tc2, Tc4 and Tc5 – produced no detectable impact on lifespan.
Future implications and medical applications
“In our tests we observed a significant advantage in terms of lifespan,” he confirms Ádám Sturm, lead author of the study. “This opens the door to myriad potential applications in the world of medicine and biology.”
That's not all: the researchers found epigenetic changes in the DNA of these worms as they aged, and in the "jumping genes". And they hypothesize that analyzing these changes could be an accurate method for determining biological age (but this will be the subject of new research).
Better understanding the pathways that control aging could lead to developing ways to extend life and improve health in our later years, researchers say. We wait and hope: Live long and prosper!


