No longer just charging and discharging: batteries are about to get a lot more interesting. Imagine a hybrid that, in addition to storing energy, produces valuable chemicals. Yes sir: a laboratory inside a battery. A dream? For nothing. A team of researchers from Tsinghua University in Beijing, China, led by Haohong Duan, is developing one which does just that.
Furfural: the perfect candidate
Furfural is an organic compound derived from various agricultural products including corn, oats and bran (the name "furfur" in Latin means "bran").
Furfural has incredible potential: it can be oxidized to furoic acid, a food preservative also useful in the synthesis of drugs and fragrances. And it can obviously also be reduced: in that case it becomes furfuryl alcohol, a precursor in resins, flavors and drugs. Chemicals that can come in very handy.
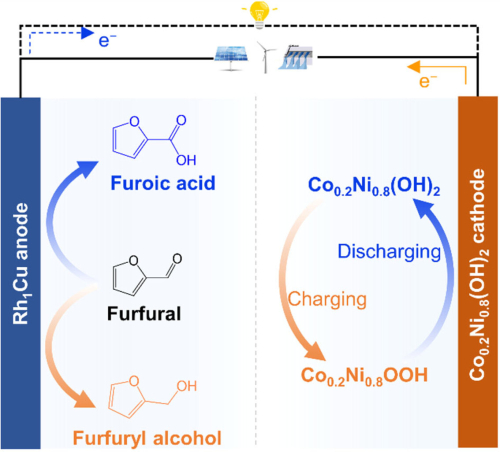
How does the hybrid battery that produces chemicals work?
The functioning of the system is a mix between the car's battery performance standard rechargeable batteries and redox flow batteries. The former store energy in their electrodes, while the latter use chemicals that oscillate between two states within the battery itself.
The Tsinghua University team combined both concepts, developing a bifunctional metal catalyst for the anode, made of a single-atom rhodium-copper alloy. During charging, this catalyst transforms the furfural-containing electrolyte into furfuryl alcohol. During the discharge cycle, however, furoic acid is formed.
A double-sided system
The result is a dual purpose battery system. After charging, four series hybrid batteries can power various devices, such as LED lights and smartphones, while simultaneously producing furfuryl alcohol and furoic acid. These chemicals are then carried away by a flow system.
Of course, the idea is still in development and there are still steps to be taken. Furfural, for example, must be continuously fed into the system and the chemicals must be separated from the electrolyte.
The study on this hybrid battery, however, (I link it here) is a really interesting step towards optimizing the efficiency and sustainability of rechargeable batteries.


