In the world of medicine, the search for an effective and safe treatment for pain has always represented a constant challenge. The recent discovery of Vertex Pharmaceuticals it could mark a significant turning point in this field. A new experimental drug, VX-548, stands out for its ability to intercept and block pain signals before they even reach the brain, promising an effective solution for moderate to severe pain without the side effects and risk of addiction associated with opioids.
Innovation in pain pharmacology: goodbye addiction
The novelty of VX-548 lies in its unique mechanism of action. Unlike opioids, which act on the central nervous system and can cause addiction and other side effects, VX-548 focuses exclusively on peripheral nerves.
These nerves, which lie outside the brain and spinal cord, are crucial in transmitting pain signals. By blocking them, VX-548 prevents pain from reaching the brain, offering relief without the downsides of opioids. It is an important perspective, if you consider the authentic massacre underway, due to the effects of drugs (like Fentanyl) also entered the drug circuit.

The mechanism of action of VX-548
The research and development of VX-548 is the result of twenty years of commitment and intense scientific research. The discovery of genetic mutations that affect pain transmission played a crucial role in the development of the drug. In particular, mutations in the Nav1.7 and Nav1.8 genes, necessary for pain transmission, have offered a new pharmacological target. VX-548 inhibits Nav1.8, temporarily blocking the protein needed for the transmission of pain signals.
Effective non-addictive painkiller: promising results from clinical trials
Vertex conducted two randomized trials to test the effectiveness of VX-548. The first involved 1.118 people undergoing abdominoplasty and The second one 1.073 people underwent surgery for hallux valgus. Both surgical procedures are commonly used in studies of people with acute, temporary pain caused by surgery.
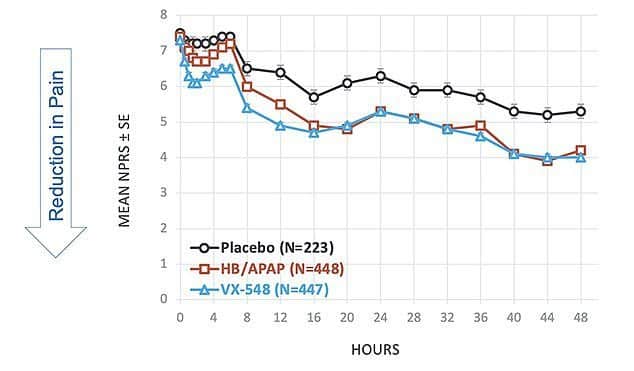
Trial participants reported a significant and clinically relevant reduction in pain, rated on a standard scale of 1 to 10.
In addition to the two efficacy studies, a third study focused on the safety and tolerability of VX-548 in individuals with various pain conditions. The results, still awaiting publication and presentation at scientific conferences, have strengthened the optimism of Vertex, which plans to request approval from the Food and Drug Administration by the middle of this year.
Future perspectives
The scope of VX-548 is currently limited to specific acute pain conditions. However, Vertex is exploring the drug's applicability in patients with chronic pain, such as that resulting from diabetic peripheral neuropathy and lumbosacral radiculopathy.
If approved, VX-548 will represent an absolute therapeutic option for the treatment of pain. An alternative to treatments such as ibuprofen, COX-2 inhibitors and opioids, the latter known for their serious risks of addiction.
With VX-548, we open up the possibility of a future where pain can be effectively managed without the risks associated with opioids. It could improve the quality of life of millions of people, and free them from opioid addiction.