A team of bioengineers at the Georgia Institute of Technology is expanding the precision and power of the therapy that is already revolutionizing oncology.
CAR-T cell therapy has been hailed by patients, clinical researchers, investors and the media as an exceptional cure for certain types of cancer.
What CAR-T therapy consists of
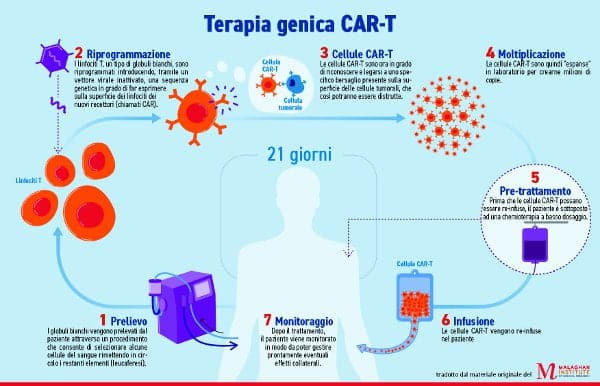
The therapy involves engineering a patient's own T cells, a type of white blood cell, in the laboratory. A chimeric antigen receptor (CAR) is then added, and finally the “personalized” CAR-T immune cells are returned to the patient's body, where they seek out and destroy tumor cells. That's how it works, when it works.
It is a new, evolving and rapidly expanding area of immunotherapy. Right now there are over 500 clinical trials around the world looking at CAR-T cells for the treatment of cancer.
CAR-T: for many, but not for everyone
CAR-T therapy has been shown to be very effective for patients with liquid cancers that circulate in the blood, such as leukemia
Gabe Kwong, Department of Biomedical Engineering at Georgia Tech and Emory.
Unfortunately, for solid tumors (sarcomas, carcinomas) CAR-T's don't work well, for several reasons. A big problem is that CAR-T cells are immunosuppressed by the tumor microenvironment.
Kwong and colleagues are making some cellular changes to improve how CAR-T cells fight cancer. They added a “genetic on-off switch” to the cells, and developed a remote control system that sends the modified T cells on “precision missions” of the tumor microenvironment, where they kill the tumor and prevent recurrence.

The team illustrated the new method in a study recently published in the journal Nature Biomedical Engineering.
The latest study
Kwong and colleagues are exploring, as mentioned, the possibility of remotely controlling cell therapies. A way to precisely “target” ai tumorswherever they are in the body. Like? activating CAR-T cells with heat when they are already inside the tumors, overcoming immunosuppression problems.
In a test on mice, researchers beamed laser pulses from outside the animal's body, where the tumor is located. Gold nanotubes injected into the tumor area transformed light waves into localized heat, raising the temperature to 40-42 degrees Celsius (104-107,6 F). It's just enough to flip the CAR-T cells' switch, but not so hot that it would damage HEALTHY tissue, or the CAR-T cells themselves.
Once “turned on,” the CAR-T cells begin to do their work, increasing the expression of cancer-fighting proteins.

An incredible genetic upgrade
The real novelty of this study, says Kwong, is the genetic engineering applied to CAR-T cells. Hard work, which he has required for the last three years.
Now CAR-T cells have a “switch” that responds to heat and other upgrades: they have literally been “rewired” to produce molecules that stimulate the immune system. And to produce them only where they are needed.
These cancer-fighting proteins are really effective, but they are too toxic to be administered systemically. With our approach we get all the advantages without the disadvantages.
Gabe Kwong
The results? Extraordinary
The latest study shows that CAR-T therapy cured cancer in mice. The team's approach not only killed the tumors, but also prevented relapses, which is critical for long-term survival.
Further studies will further delve into the customization of CAR-T cells and how heat will be radiated to the tumor area.
“On humans we will use focused ultrasound, which is absolutely non-invasive and capable of affecting any site of the body,” says Kwong. “One of the limitations of the laser is that it doesn't penetrate very far into the body. In case of deep malignancy, this is a problem. We want to eliminate problems."