Researchers from the Reik laboratory at the Babraham Institute in Cambridge have achieved excellent results. Using the four Yamanaka reprogramming factors (OSKMs) they epigenetically rejuvenated human cells by 30 years.
Previous experiments had failed in one element. While exposure of human cells to Yamanaka factors rejuvenates them, it induces pluripotency to transform them into stem cells, causing them to lose their cellular identities (and therefore function).
It's a long-standing problem. You need to expose your cells to these factors long enough to get the rejuvenation, but allowing them to retain their identity.

The Yamanaka factors
There are four transcription factors: Oct4, Sox2, Klf4 and cMyc (OSKM). Using them reliably creates iPS cells, but can cause unwanted effects, some of which can lead to cells becoming cancerous.
The Cambridge study of human cells
The researchers of this study they used an approach that exposed the cells to sufficient reprogramming factors to push them beyond the limit where they were considered somatic rather than stem cells. Just beyond. Fibroblasts that have been reprogrammed in this way have retained enough of their epigenetic cellular memory to become fibroblasts again. Researchers call this new method as transitional reprogramming in the ripening phase (MPTR).
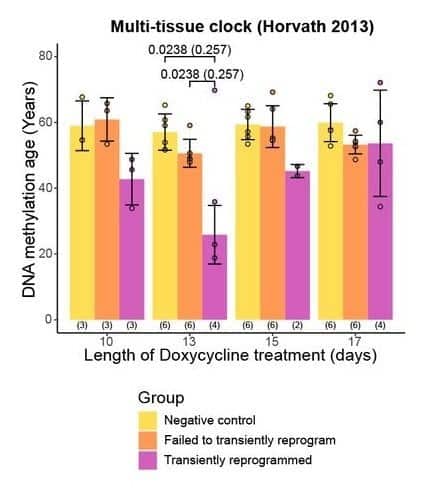
Great results, and some drawbacks
The MPTR method has had substantial positive results. According to Horvath's multi-tissue clock, a 2013-born biochemical test used to measure age, after 13 days of reprogramming, 60-year-old human cells became epigenetically equivalent to cells that were around 25 years old. Another test born in 2018, the Skin and Blood Epigenetic Clock, showed that cells that were around 40 years old were epigenetically reverted to those of a 25-year-old. The technique also substantially rejuvenated the transcriptome, the collection of proteins produced by genes.
There are, of course, some caveats. The most important, of course, is that this experiment was performed on human donor cells, but not on a human volunteer. Therefore, systemic factors known to influence the epigenome, such as those found in ancient blood, were not applied.
PMTR on human cells: 10 days are few, 17 are too long
The exposure of these cells to Yamanaka OSKM factors was also controlled in the dosage forms. 10 days of exposure did not epigenetically rejuvenate the cells as well as 13 days of exposure, but the researchers showed that too much exposure (15 and 17 days) led to cellular stresses that re-aged the epigenome. This study had only a few donors and the results after 13 days varied greatly from person to person.
The effect of MPTR exposure on telomeres
MPTR did not positively influence the aging sign of telomere attrition. When the cells were allowed to be completely reprogrammed into stem cells, their telomeres began to extend; but this partial reprogramming led to a moderate shortening of telomeres even though it rejuvenated the epigenomes of the cells.
Furthermore, MPTR did not work on all human cells and achieved these results after screening procedures that divided the cells into failed and successful reprogramming groups. However, even the “failed” group achieved partial successes in many key parameters of aging and cellular health.
Conclusions
Although this experiment demonstrated that it is possible to epigenetically reprogram viable human cells under laboratory conditions, applying such an approach in the clinic would require considerable development of the biotechnological foundations in order to provide each of the patient's individual cells the exact amount of OSKM it needs to be successfully rejuvenated and no more. This technology is not yet on the horizon.
And for therapies based on human cell cultures?
The consideration is different as to whether such an approach can be used for the development of human cell cultures to be reintroduced into an elderly person. This experiment used fibroblasts, which form collagen, so it is reasonable to imagine a world in which such reprogrammed human cells are developed as a therapy against wrinkles and other effects of aging on the extracellular matrix.
This approach could one day be used to create viable, rejuvenated populations of muscle (including heart muscle) and brain cells. Such newly reprogrammed “quasi-somatic” human cells could ultimately be the best option in many clinical applications.
Whichever approach proves most effective, we look forward to the day when our cells can be epigenetically reprogrammed in youth and reintroduced into our bodies to ward off the signs of aging.


